
What are LSE plastics and HSE plastics?
Each type of material including plastics has specific surface energy. It quantifies the disruption of intermolecular bonds that occurs when a substrate is created. Solid materials such as plastics are less energy consumable which means that the molecules on the surface have more energy compared with the molecules in depth. According to the surface energy of the molecules on the surface, plastics are divided into two types – with a low energy surface (LSE plastics) and with a high energy surface (HSE plastics).
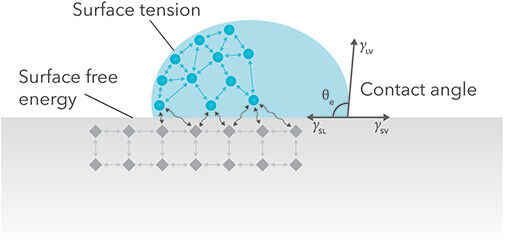
Surface energy influences adhesion as defining the ability of adhesives and tapes to “wet out” plastic surfaces. LSE plastics are difficult to bond because their molecules on the surface has low energy and can not join with these of the adhesive. Unlike LSE plastics, HSE plastics are easy to bond. On their surface there are molecules with high energy which join easier to molecules of the adhesive.
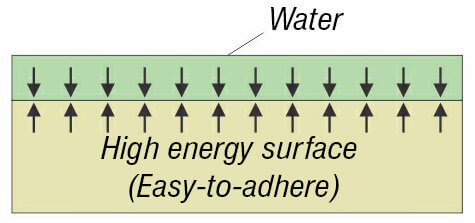
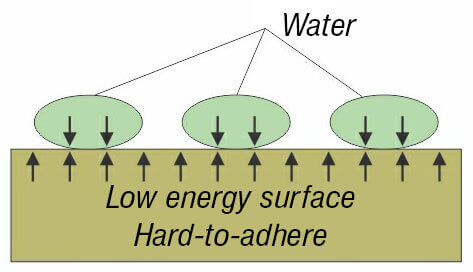
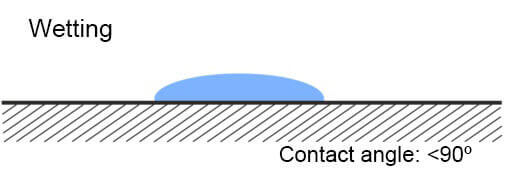
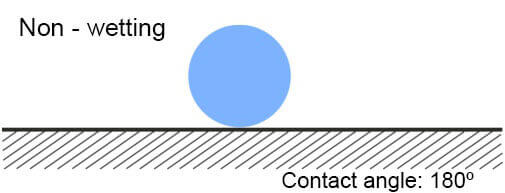
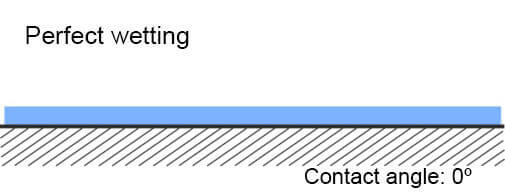
The most simple way to recognize what is the surface energy of the plastic – low or high, is called Wetting Method. When water drops are sprinkled on the plastics and they spill on it, this means that the plastic has high surface energy. If drops do not spill on the plastic (or another solid material), it has a low energy surface.
I. Wetting
II. Non-wetting
I. Contact angle graphic
The most used LSE plastics are:
- Polymethyl methacrylate (PMMA) – widely known as plexiglass. It is used in applications similar to these of glass. But PMMA is preferred in many industries because of its good price, easier processing, efficiency, and visible light transmission. It is widely spread in secondary industry, interior decorations, and advertising;
- Polypropylene (PP) – It is used for bottles caps, drinking straws, yogurt pots, kitchen appliance parts, car parts (bumpers), plastic piping systems;
- Polyethylene(PE) – from this plastic are produced shopping bags, garbage bins, and more than 30% of the kids’ toys worldwide;
- Polystyrene, known as styrofoam – widely used in construction, for thermal insulation. Kids’ building blocks, plastic utensils, food packs are made of polystyrene too.
- ЕVA (porous rubber) – it is foam polyethylene. For producing of yoga mats, kids toys, fishing equipment, EVA clogs, and anatomic insoles; in the construction sector – for different types of insulation;
- Polycarbonate (PC) – used for CDs, sunglasses, police shields, “armored” windows;
- ABS (acrylonitrile butadiene styrene ) – a thermoplastic used for panels for electronics ( monitors, printers, keyboards), pipes, LEGO blocks.
The most used HSE plastics are:
PVC (extremely soft), cast plastics, ABS mixtured with PC, rubber, ЕPDM, HDPE, PET/PETG, polyamide, TPOs, powder-coated surfaces.
- АBS mixtures with polycarbonate – for producing car interior parts, panels for mobile phones.
- HDPE (polyethylene with high density) – for manufacturing bottles of liquid cleaners, milk packs, and others.
- PVC (Polyvinyl chloride) – for the production of sewer pipes and gutters, bathroom curtains, window frames, floorings, and even gramophone plates. Most of the bathroom furniture is made of PVC.
- PET/PETG (Polyethylene terephthalate) – a part of polyesters group. About 60% of the world’s production of this polymer is used to make yarn from which are made clothes, sails, and many others.
- ЕPDM (synthetic rubber) – it is used to manufacture rubber membranes for hydro insulation, car rubber sealings, garden hoses, cable insulation, and more;
- TPOs ( thermoplastic polyolefin ) – are a combination of polypropylene and ethylene-propylene rubber. It is used for producing multilayer thermally films for the packaging of food products.
- Полиамид (Nylon) – for manufacturing of sliding bearings and strips, bushings, gears, nuts and bolts and more for automotive and machine industries; in the textile industry to replace the silk in clothes producing.
These plastics must be extremely clean before bonding. The right choice of adhesive is responsible for only 50% of the adhesion strength. The other 50% is responsible for the application method.
How to prepare HSE plastics and LSE plastics for bonding?
The preparation for bonding is very importance for both types plastics of LSE and HSE. Bonding of plastics with low surface energy may require to use a primer or additional preparations.
STEP 1
Degrease with liquid from IPA / H2 O – isopropyl alcohol.
STEP 2
Make the surface rugged (if it is possible, and for opaque details). It enlarges the bonding area and removes oxidation.
STEP 3
Degrease again with isopropyl alcohol.
STEP 4
Applying a Primer: when need to bond LSE surfaces, glass, powder-coated surfaces, and plastics (soft and cast). The primer seals the surface and prepares it for bonding.






