

Plastics of all shapes and types have become indispensable in our everyday lives. If you look around, you will find that an increasing number of natural materials are being replaced by plastics. This has resulted in many technical challenges, one of the biggest being bonding.


There are numerous, well-known benefits of abandoning the use of metal in favor of plastics. Using plastics brought about significant changes in the simplicity of maintenance, as well as durability of products. This has in turn made life easier for users on a level unattainable in the past, when metals were used for the same products. Let us use our boats and their parts as an example. In most cases, plastic is lighter than metal, which means that the final product made from it is lighter as well, which makes quite the difference when you are at sea.

Other characteristics of plastic materials include resistance to corrosion and chemicals. The last, and perhaps the most important aspect is their price. Today, plastic parts are mostly mass produced from molds, which significantly reduces the price of the most expensive work component, human labor. But the area where every single boatsman has noticed the greatest improvement is precisely corrosion resistance. Plastic vessels do not rust, and plastic tanks will not be damaged by the corrosive fuel and become covered in rust.
There is no need to paint them over, to fight corrosion, no need to worry about grounding, zincing… Simply put, there is no need for maintenance!
Things are great when maintenance isn’t necessary, but what happens when something needs to be repaired?
In this regard, plastic components proved much more complicated in some cases. Unlike metals, which can simply be repaired by welding, riveting, or adding screws, plastic materials could not be repaired as easily. The only remedy for this has been chemical compounds such as adhesives, while some plastic materials, like polyethylene, could be welded, but only by a very complex process. The low purchase price, as well as the high costs and complexity of repairs, have to lead to these materials still being seen as disposable. Even when it came to valuable objects that were truly worth the effort and that should have been repaired when any kind of damage appeared.

One example is the polyethylene tanks on our boats and even entire boats such as Whally or most of the sit-on-top kayaks that can be seen along the coastline… All of these are valuable, and it would be a shame to write them off simply because they have been damaged.
Unfortunately, this was often the case.
Despite their excellent mechanical properties, durability, and resistance to mechanical, as well as most other stresses, polyethylene parts also sustained damage, and among all plastic materials, these were the most difficult to repair.
Up until now, damage to components made from polyethylene and similar plastic materials meant one of two things: the object would either be welded if it had thicker walls, or it would be disposed of. Bonding was out of the question. Until now…
Why are some plastic materials so difficult to bond?
Most of us have at some point in our lives experienced the need to bond things together that simply would not bond. All of the adhesives that were ideal for bonding metal, leather, ceramics, and even some types of plastic, proved completely ineffective on some surfaces. What was the problem? All of these surfaces that are difficult to bond have the same property – low surface energy. Surface energy is energy created as a result of the materials’ surface tension.
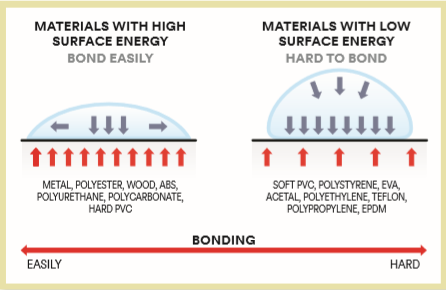
The stronger the interaction between atoms on the surface, the higher the surface energy. What is interesting is that the surface energy of solid surfaces cannot be measured directly, but can be determined by a very simple empirical method – the so-called wetting method. The experiment is simple. A drop of water is placed on the surface and the contact angle between the drop and the surface is measured. The larger the angle, the lower the energy.
When it comes to polyethylene, the drop will take the shape of a bead whose wall will form an angle of 72º with the surface. When it comes into contact with glass, the same drop will spread over a much larger surface, and its edge will rise to an angle of 5º relative to the surface.
Experience shows that, on the molecular level, these materials behave in the same manner as other liquids as well. In other words, the contact surface between the adhesive and low surface energy material will be limited, as will be their adhesion.
Many call this low surface energy effect the “lotus effect”, although us shoremen are more familiar with collard greens, from whose surface water, paint, and other liquids simply slide off, leaving no residue. Thermoplastic olefin (TPO), polypropylene (PP), and high-density polyethylene (HDPE) exhibit the same property.






